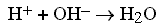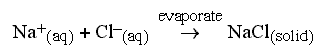|
Many industrial processes consist of
equilibrium reactions.
We all know that if a simple acid and base are mixed, the result is the
formation of water as an ionic reaction:
If the water is removed by evaporation, then salt is left. For example,
if one mixes solutions of sodium hydroxide and hydrochloric acid, the
hydrogen ions and hydroxide ions form water, leaving free sodium ions and
chloride ions in solution. After evaporation, these crystallise to form
sodium chloride:
Certain organic compounds exhibit analogous reactions, in particular the
elimination of water in the elimination reaction between an amine and an
organic acid:

Copyright © 1999-2002 Key Centre for Polymer Colloids.
Last Modified: Thursday February 15, 2001
|
|
|
|