
More About Nylon66
 |
 |
 |
|
 |
|
 |
|
 |
|
 |
|
 |
|
 |
|
 |
|
 |
|
The product that comes out of the nylon rope trick, or by condensing the acid and amine together under ambient conditions, is not very satisfactory as a material: it is very weak, and has a low melting point. This is because its molecular weight is not very high (although one says that polymers are molecules with "infinite" molecular weights, in fact their molecular weights are high but not infinite). Carothers found that the physical properties (melting point and strength) of the product were improved by first forming the salt of the amine and acid:
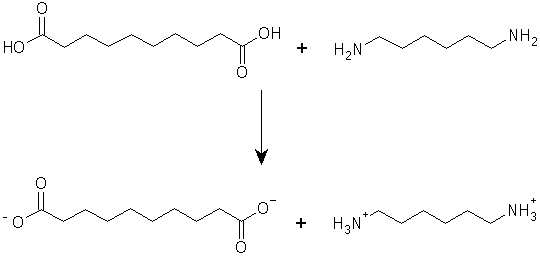 |
and then heating this salt:
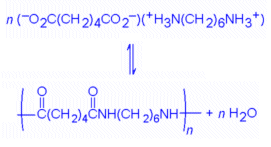 |
The product has same chemical formula as that given previously for nylon, but has a higher molecular weight.
Equilibrium and nylon
This reaction is an equilibrium one, and at ordinary temperatures lies to the left. To obtain a high yield, it is necessary to drive the reaction to the right. The reaction is endothermic. Hence by Le Châtelier's Principle, the way to drive it to the right is to heat it. Again using Le Châtelier's Principle, one can also drive the reaction to the right by removing product, in this case the water that is formed by the elimination reaction. A high yield can be obtained by running the reaction at 200 °C and letting the water be removed as steam.
Note: you may wonder at this point why it is that your nylon clothes do not fall apart when ever they get wet (as implied by the equilibrum favouring the reactant side). This is due to the kinetics of the reaction. The rate of the reaction is extremely slow, so even if the equilibirum would like to favour the reactants, it will take longer than the time you are wearing nylon to actually do so.
 |
 |
 |